Faculty and students from Utah’s BME department joined over 5500 registered attendees at the annual Biomedical Engineering Society (BMES) national meeting from October 8-12, 2025 in San Diego, CA. They enjoyed a rich, diverse technical, educational, industrial, and policy-related biomedical engineering program that featured topical, special, plenary and keynote sessions along with award forums and education and technology sessions in all-day parallel session formats. Industrial speakers, AIMBE sessions, and special topics panels and workshops also highlighted the annual event.
Seven Utah BME faculty and 17 Utah BME undergraduate and graduate students attended to present their research and participate in networking opportunities with other participants in the BME community. Utah BME staffed an exhibit booth on the busy BMES exhibition floor with over 100 other academic programs and relevant BME sponsors and partners.
Utah BME students presented the following posters or talks:
Utah BME undergraduate student presenters:
Connor Browning (co-authors: Lindsay C. R. Tanner, Anna Busatto, Ben Orklid, Eric Paccione, and Rob S. MacLeod)
Faculty Advisor: Rob S. MacLeod
Title: Decoding the Spatiotemporal Patterns of Myocardial Ischemia
Abstract: Cardiovascular disease is the leading cause of mortality, accounting for 20.5 million deaths worldwide in 2021. The most common form of cardiovascular disease is coronary artery disease (CAD), with 98% of the world’s population having at least one CAD risk factor. CAD results in a limited flow of blood and oxygen to the myocardium and ultimately can lead to myocardial ischemia. The standard clinical method for diagnosing myocardial ischemia is through the examination of a 12-lead ECG. However, ECG diagnosis has a sensitivity of 50%-70% and a specificity of 70%-90%. This poor performance by the ECG can lead to potential misdiagnoses with severe consequences, underscoring the need for improved diagnostic methods.
Kiley Cole (co-authors: Monika K. Buczak, Haohan Zhang, Jacob A. George)
Faculty Advisor: Jacob A. George
Title: A Low-Profile High-Density Electromyography Neckband for Recording Neck Muscle Activity
Abstract: The predominant use of electromyography (EMG) with the extremities has led to specific formfactors conducive to the arms and legs. Here, we describe the design and validation of a new wearable for recording EMG from the neck. EMG from the neck is useful for intraoperative neuromonitoring, outpatient monitoring of disease progression, and control of assistive technology. The current approach of using adhesive electrodes is time consuming and not practical for extended at-home use. Here, we introduce a low-profile high-density EMG neckband that supports the unique requirements of the neck. The EMG neckband ensures broad muscle coverage across a range of neck sizes without hindering neck mobility or function (e.g., breathing, eating, speaking). Relative to the clinical and research standard of adhesive electrodes, the EMG neckband provides significantly faster donning and doffing times (seconds instead of minutes) and comparable signal quality and myoelectric control. This work constitutes an important step towards the translation of neck EMG as an assistive and diagnostic wearable, which in turn may improve quality of life for individuals with neuromuscular impairments.
Landon Deroche
Faculty Advisor: Rob S. MacLeod
Title: Comparing Rule-Based to Experimental Coronary Arteries
Abstract: Cardiovascular disease is the number one cause of death in the world, with myocardial ischemia (MI) being one of its most serious forms. MI occurs when reduced blood flow deprives the heart muscle of essential oxygen and nutrients, often resulting in life-threatening consequences. Clinically, the electrocardiogram (ECG) is used for diagnosing MI; however, its limited sensitivity and specificity may lead to misdiagnosis and suboptimal treatment. One approach to improving diagnostic accuracy is to capture the detailed structure of the coronary vasculature, as the coronary arteries play a central role in myocardial health. Capturing this information is enabled by recent advances in computed tomography (CT) imaging to noninvasively visualize these blood vessels. However, CT angiography typically captures only larger vessels due to spatial resolution limits. To overcome these challenges, computational methods that generate rule-based estimates of the vasculature have been utilized. In this project, we compare experimentally acquired coronary artery networks from high-resolution CT images with computer-generated vasculature, aiming to identify similarities, differences, and potential improvements in modeling techniques. This project is intended to ultimately refine noninvasive diagnosis and enhance our understanding of MI pathophysiology.
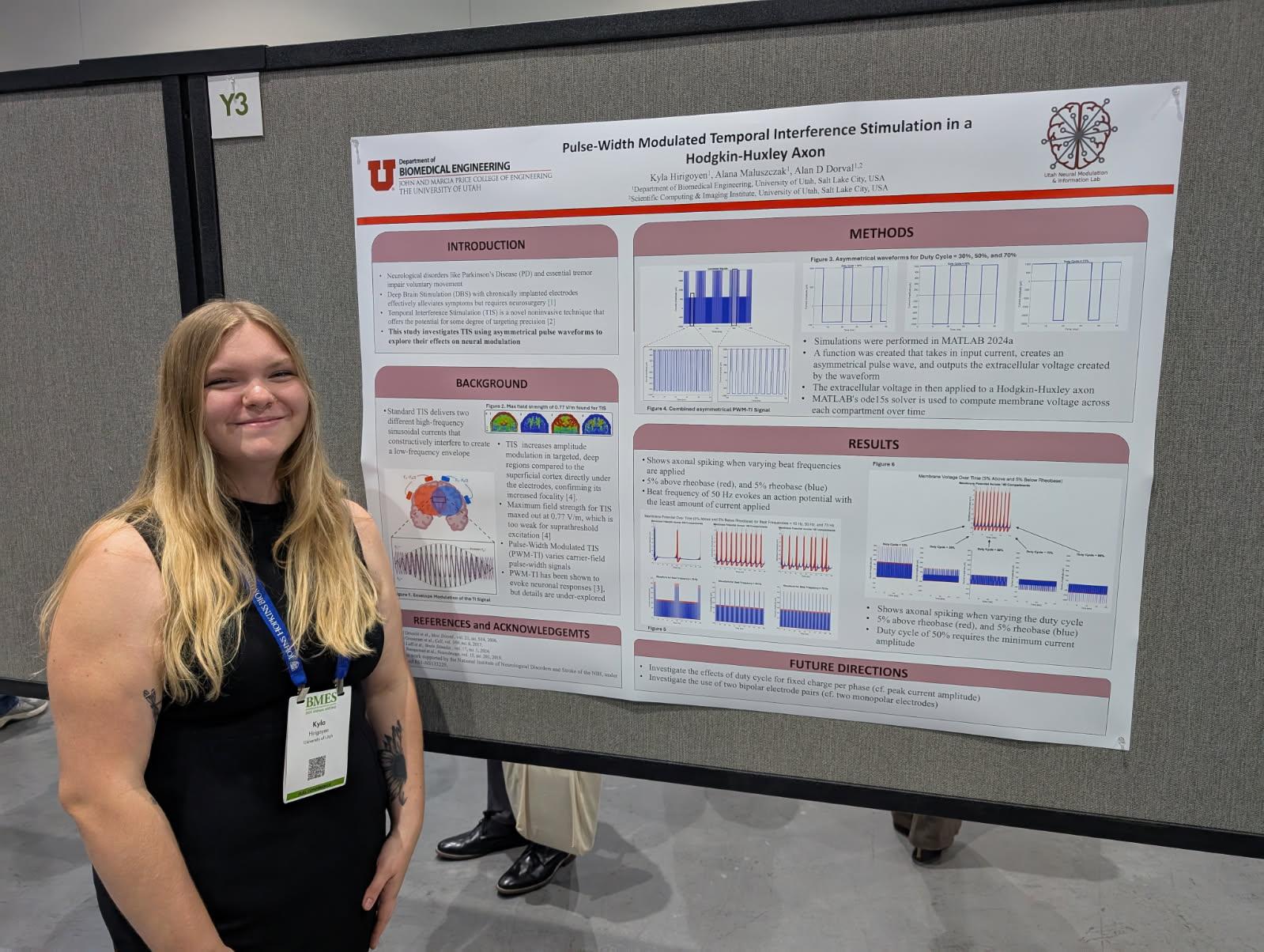
Kyla Hirigoye (co-authors: Alana Maluszczak, Alan D. Dorval)
Faculty Advisor: Alan D. Dorval
Title: Pulse Width Modulated Temporal Interference is a Hodgkin-Huxley Axon
Abstract: Electrical neuromodulation from electrodes chronically implanted into specific deep brain regions alleviates symptoms for a variety of neurological disorders. However, implantation requires neurosurgery, which adds substantial expense and risk of complications. Noninvasive neuromodulation techniques offer an alternate approach to neurological-symptom alleviation that can avoid the limitations associated with chronic implantation. However, these noninvasive techniques generally lack the targeting focality of invasive approaches. With insufficient targeting focality, noninvasive approaches can affect either too little of the deep brain target to alleviate symptoms, or too much surrounding neural tissue, which drives neurological side-effects.
Temporal interference (TI) stimulation is a relatively novel noninvasive neuromodulatory technique that enables some notion of targeting focality. Standard TIS applies two high-frequency sinusoidal currents from two distinct contact pairs at slightly different frequencies. The two sinusoidally varying electric fields interfere constructively and destructively; their combination creates a high carrier frequency field at the average of the two frequencies, modulated by a low beat frequency field at the difference of the two frequencies. Preclinical efforts over the past decade have demonstrated neuromodulatory effects at the beat frequency, but the sinusoidal field amplitudes required to elicit a response are not clinically viable in human patients.
Utah BME graduate student presenters:
Priyanka Arunachalan (co-authors: Hayrettin Tonbul, Cansu Urman Tunc, Nitish Khurana, and Hamidreza Ghandehari)
Faculty Advisor: Hamidreza Ghandehari
Title: Synthesis and evaluation of cellular toxicity of PEG conjugated silica nanoparticles for drug delivery applications
Abstract: Silica nanoparticles are among the diverse nanomaterials being explored for drug delivery applications due to their chemical stability, favorable biocompatibility, and tunable porosity. To enhance their circulation time and reduce recognition and clearance by the mononuclear phagocyte system (MPS), silica nanoparticles can be surface functionalized with poly(ethylene glycol) (PEG). In this study, we synthesized both porous and non-porous PEG-conjugated silica nanoparticles. We assessed their viability, potential to induce reactive oxygen species (ROS), and cell cycle phase distribution using RAW 264.7 murine macrophages as a model cell line.
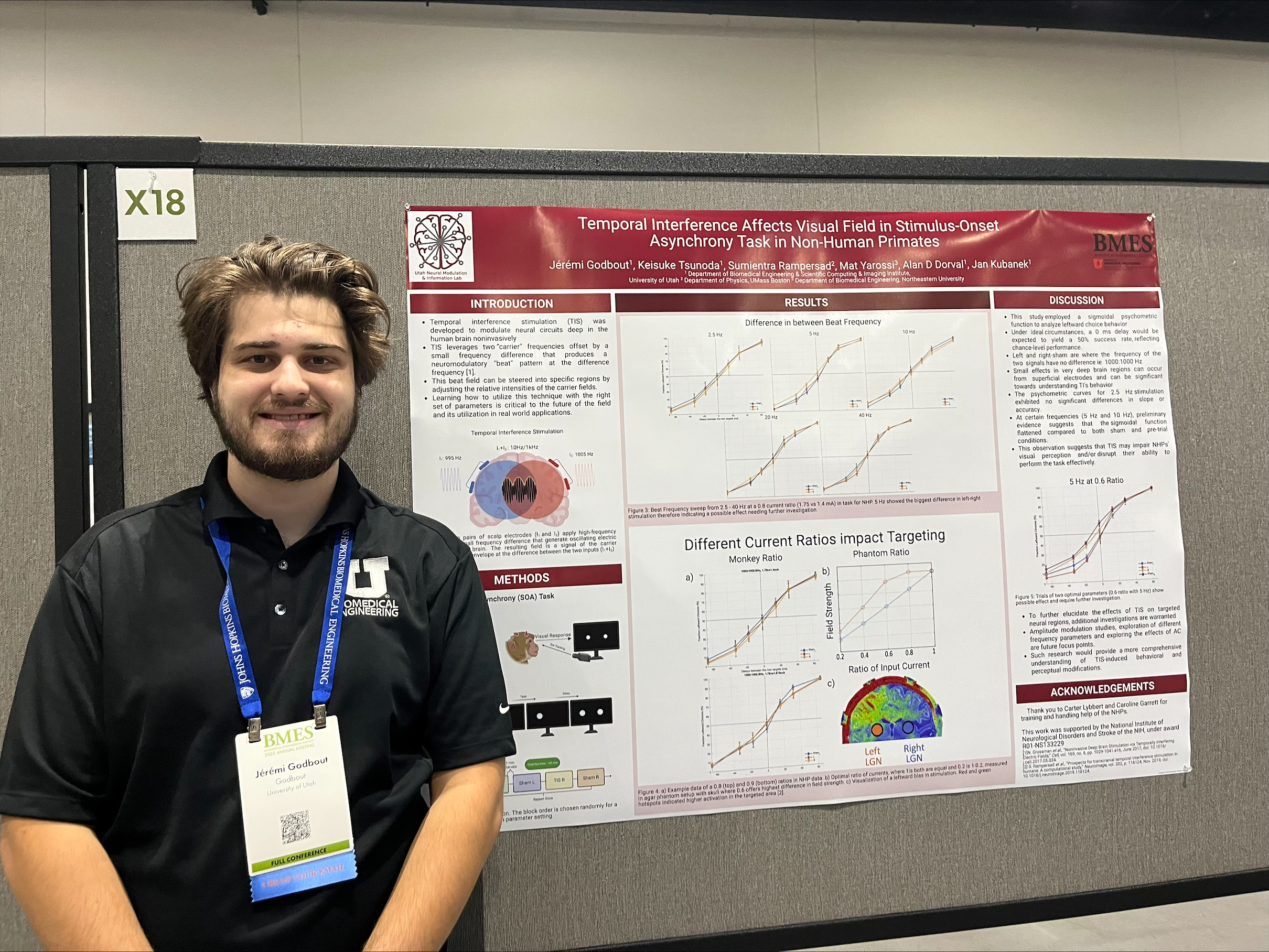
Jérémi Godbout (co-authors: Keisuke Tsunoda, Alan D. Dorval, and Jan Kubanek)
Faculty Advisor: Alan D. Dorval
Title: Temporal Interference Affects Visual Field in Stimulus-Onset Asynchrony Task in Non-Human Primates
Abstract: Temporal interference stimulation (TIS) was developed to modulate neural circuits deep in the human brain noninvasively, as an alternative to traditional transcranial brain stimulation. Existing clinical transcranial neuromodulation — such as transcranial alternating current (tACS), direct current (tDCS), and magnetic (tMS) stimulation — share a common fundamental limitation: a trade-off between brain depth, neural target focality, and activating field intensity. The TIS approach has the potential to circumvent this trade-off, enabling deep and focal neural activation. TIS leverages two “carrier” frequencies offset by a small frequency difference that produces a neuromodulatory “beat” pattern at the difference frequency. This beat field can peak deep in the brain, can be more focal than conventional stimulation, and can be steered into specific regions by adjusting the relative intensities of the carrier fields. Learning how to utilize this technique with the right set of parameters is critical to the future of the field and its utilization in real world applications.
Md Mahedi Hasan (co-authors: Kaghoon Choi, Tamanna Islam, Jungkyu Kim, and Taslim Al-Hilal)
Faculty Advisor: Taslim Al-Hilal
Title: Inflatable Chip to Study Malignant Ascites in Ovarian Cancer
Abstract: A central obstacle in ovarian cancer (OC) disease is the lack of advanced bioengineering models that can accurately mimic intraperitoneal pressure fluctuations and their effects on cell behavior within the peritoneal cavity. OC is the deadliest gynecological malignancy, primarily because of its aggressive peritoneal spread and frequent development of malignant ascites, both of which are closely associated with poor prognosis and limited treatment success. Existing in vitro systems fall short in recreating the dynamic and complex tumor microenvironment, while in vivo models are constrained by the inability to conduct real-time analysis or precisely control site-specific ascites accumulation, limiting both mechanistic insights and the development of targeted therapies. These shortcomings impede the detailed investigation of metastatic mechanisms and hamper the evaluation of interventions directed at the peritoneal microenvironment. Microfluidic platforms offer physiologically relevant multi-cellular co-cultures, precise microenvironmental control, and real-time monitoring of key disease processes. The Inflatable chip to study Malignant Ascites in ovarian Cancer (iMAC) chip, with its integrated simulation of vascular, mesothelial, and tumoral compartments alongside mechanical actuation of ascites-driven peritoneal inflation, represents a significant improvement over previous models, enabling unprecedented spatiotemporal control and physiological relevance in modeling OC metastasis and malignant ascites formation.
Jonah Hollbrook (co-authors: Christian Lewis, Hunter Levis, and Robby D. Bowles)
Faculty Advisor: Robby D. Bowles
Title: DNA Damage in Intervertebral Disc Degeneration: Quantification and Modulation with BLST
Abstract: Low back pain is the world’s leading cause of years lived with disability. As such, low back pain creates a tremendous economic burden of over $100 billion in the United States annually. The leading cause of low back pain is degenerative disc disease (DDD), accounting for 40% of all instances. The pathophysiology of DDD has been extensively characterized. In a degenerative intervertebral disc (IVD), the pH decreases from 7.1 to below 6.2 in severely degenerated discs. Further, cells in degenerative discs begin to exhibit signs of senescence. Cellular senescence is a well understood biological phenomenon in which cells enter a state of cessation of growth and division. Cells in a senescent state often exhibit the senescence-associated secretory phenotype (SASP), identified by the production of proinflammatory signals, which ultimately leads to degradation of surrounding tissue and subsequent pain and disability. Further, cellular senescence has been postulated to be a response to DNA damage, but the exact mechanism of this response remains unknown. The gene ZNF865 (BLST) has recently been identified by the Bowles lab as a regulator of cellular senescence with involvement in intervertebral disc degeneration; however, its functional mechanism and interactions have not been extensively characterized. Here, we investigate differences in DNA damage in healthy and degenerative human intervertebral disc cell populations as well as healthy cell populations subjected to low pH conditions. Further, we quantify differences in DNA damage upon altering expression of BLST with CRISPR regulation techniques.
Tamanna Islam
Faculty Advisor: Taslim Al-Hilal
Title: Bioengineered Approach to Study Cell-specific Durotaxis in Lung Fibrosis and Pulmonary Arterial Hypertension
Abstract: Durotaxis, the directed migration of cells along gradients of matrix stiffness, has emerged as a critical mechanobiological process in the pathogenesis of multiple fibrotic and degenerative diseases. This mechanosensing phenomenon represents a fundamental yet underexplored mechanism underlying tissue remodeling in chronic diseases. We have recently identified this novel mechanism in which pulmonary cells, including fibrotic fibroblasts and diseased smooth muscle cells (SMCs), are recruited to sites of tissue injury via “durotaxis”, a form of directed cell migration driven by spatial gradients of matrix stiffness. Here, we explore the molecular mechanisms driving cell-specific durotaxis in the pathogenesis of idiopathic pulmonary fibrosis (IPF) and pulmonary hypertension (PH), both of which are characterized by the presence of steep stiffness gradients and fibrotic destruction of the lung parenchyma, and develop tools to address the lack of suitable models to comprehensively study durotaxis in vitro and in vivo.
Christian Lewis (co-authors: Jake Polaski, Brandon Lawrence, Jay Gertz, and Robby D. Bowles)
Faculty Advisor: Robby D. Bowles
Title: CRISPRa Regulation of ZNF865 Decreases SASP Expression and Reverses Chromatin Differences Between Healthy and Degenerative NP cells
Abstract: Pathology of the intervertebral disc (IVD) and associated back pain is a major healthcare concern in the US. About 40% of this back pain can be attributed to degeneration of the IVD. Degenerative disc disease (DDD) has been closely associated with several hallmark phenotypes, relating to increased inflammation and senescence in degenerating discs. However, there is a lack of information regarding the major genetic determinants of IVD disease onset and progression. Therefore, determining the underlying molecular etiology of IVD pathogenesis has the potential to reveal novel therapeutic opportunities, which would address a critical need for the development of targeted treatments for IVD degeneration. Recently, in a set of CRISPRa genome-wide screens, our lab identified a previously uncharacterized zinc finger protein, ZNF865, also known as BLST, that produces robust cell-engineering phenotypes relating to cell cycle, protein processing, and cellular senescence. Preliminary data from publicly available RNAseq datasets suggests that ZNF865 is differentially regulated within degenerating human IVDs. Here we investigate the molecular phenotypes associated with CRISPRa of ZNF865 within primary human IVD samples and examine differences in chromatin accessibility between healthy and degenerative samples.
Alana Maluszczak (co-authors: Mathew Yarossi, and Alan D. Dorval)
Faculty Advisor: Alan D. Dorval
Title: Exogenous inputs that scale ion channel kinetics modulate activity in a computational CA3 pyramidal cell
Abstract: Exogenous inputs such as heat, mechanical vibration and pressure, electromagnetic radiation, and pharmacological agents have been shown to affect ion channel kinetics and modulate neuronal activity. Neuronal ion channels comprise transmembrane proteins with gating domains that open and close a central pore through which specific ions tunnel across the cell membrane. The opening and closing of these ion channel gates are stochastic processes with rate constants fit to experimentally derived functions of membrane potential, calcium concentration, and other physiological variables. However, the quantitative effects of the aforementioned exogenous inputs on the rate constants, and the modifications to neuronal behavior that they can drive, are poorly understood. In this work, we seek to categorize how relatively minor changes in ion channel rate kinetics can substantially modulate neuronal behavior and lead to qualitative changes in response dynamics. To investigate changes that increase neuronal excitability, we focus on ions with depolarizing effects: sodium and calcium. We aim to develop a mechanistic understanding of how ion-channel responses to exogenous stimulation promote qualitative changes in neuronal spiking patterns, synaptic plasticity, and the overall behavior of the neuronal network activity.
Nejra Mujkanović (co-authors: Chelsea E. Pollard, Rachel Detwiler, Philip Moos, Jeremiah A. Alt, Gretchen M. Oakley, Richard R. Orlandi, Kristine A. Smith, Abigail Pulsipher, and Hamidreza Ghandehari)
Faculty Advisor: Hamidreza Ghandehari
Title: Sinonasal Tissue Microenvironment Characterization in Eosinophilic Chronic Rhinosinusitis to Inform Drug Delivery Strategies
Abstract: Chronic rhinosinusitis (CRS) is a heterogeneous inflammatory disease of the sinonasal cavity, impacting approximately 12% of adult Americans. This condition significantly impacts patients’ quality of life and places a substantial burden on the U.S. healthcare economy. One of its endotypes, eosinophilic chronic rhinosinusitis (eCRS), affects roughly 4% of patients and presents ongoing challenges in medical and surgical management. eCRS is primarily characterized by high eosinophil counts, nasal polyps (NP)—non-cancerous growths, and eosinophil-associated type 2 inflammatory pathways that contribute to sinonasal tissue matrix destruction and mucus hyperviscosity.
Despite current treatment options—including saline rinses, intranasal corticosteroid sprays, oral corticosteroids, antibody biologics, and surgery—nearly half of eCRS patients remain unresponsive. These treatments are hindered by limitations such as poor tissue penetration against inflamed anatomical barriers, systemic side effects from oral corticosteroids, and multiple revision surgeries. There is a critical need for safe and effective localized therapeutic strategies underlying tissue damage and inflammation seen in eCRS.
Comprehensive profiling of the environment is essential for developing localized targeted therapies in reducing severe inflammation in eCRS. We hypothesize that epithelial damage and immune dysregulation contributes to chronic inflammation in eCRS, impeding progress in localized delivery of therapeutics. This study aims to characterize the epithelial cell changes, distribution of type 2 immune cells, and viscoelastic properties of mucus in eCRS.
Sofia Ruiz Castillo (co-authors: Muhammad S. Khan, Kyoichiro Yazaki, Emmanuel Offei, Aba Vaziri, Ankur Shah, Douglas Smego, Ting Ting Hong, Robin M. Shaw, and Derek J. Dosdall)
Faculty Advisor: Derek J. Dosdall
Title: Cardiac Bridging Integrator 1 Gene Therapy Reduces Fibrosis in Failing Myocardium in a Pre-Clinical Model of Ischemic Heart Failure
Abstract: Heart failure (HF) is a significant health concern, impacting over 6 million individuals in the United States. A common cause of HF is myocardial infarction, which leads to scar formation in the ischemic zone and increased stress and remodeling in the rest of the myocardium. This remodeling is not confined to the ischemic area; hormonal and paracrine signals, along with increased mechanical stress, contribute to reactive fibrosis in regions outside of the infarcted zone. The presence of scar tissue and interstitial fibrosis adversely affects cardiac function both mechanically and electrically. Cardiac bridging integrator 1 (cBIN1) is a membrane scaffolding protein that has been proposed as a therapeutic target for HF. cBIN1 is crucial for organizing the calcium handling machinery in myocardial membranes, and its levels are reduced in HF. This reduction leads to calcium mishandling, a key characteristic of the disease that contributes to organ dysfunction. Exogenous cBIN1 gene therapy has been tested in a preclinical large animal model of ischemic HF, showing improvement in left ventricular (LV) ejection fraction (EF) compared to control therapy. However, it remains unclear whether this mechanical improvement affects tissue remodeling in other areas of the LV. The aim of this study is to investigate fibrotic deposition outside of the infarct zone after gene therapy in a pre-clinical model of ischemic HF.
Sushanto Kumar Saha (co-authors: Cansu Umran Tunc, Nitish Khurana, Philip J. Moos, Hamidreza Ghandehari
Faculty Advisor: Hamidreza Ghandehari
Title: Modulation of cell signaling pathways in silica nanoparticle-saturated macrophages
Abstract: In recent years, silica nanoparticles (SNPs) have been investigated for delivery of bioactive agents1-3. When systemically delivered, a significant amount of silica nanoparticles are taken up by the macrophages of the reticuloendothelial system. Depending on the dosage administered, the macrophages can become saturated with nanoparticles, potentially impacting their cell signaling and the overall immune system function. In this study, we used nonporous, mesoporous (MSNPs), and hollow-mesoporous silica nanoparticles (HMSNPs) to study the gene expression alterations and modulation of cell signaling pathways upon saturation of RAW 264.7 murine macrophages with nanoparticles.
Ata Ullah (co-authors: Matt D. Nelson, Bruce K. Gale, and Taslim Al-Hilal)
Faculty Advisor: Taslim Al-Hilal
Title: 3D-Printed Lung Microphysiological System for Cancer Studies and Drug Testing
Abstract: Polydimethylsiloxane (PDMS) has revolutionized Organ-on-Chip technology due to its excellent optical transparency, gas permeability, and ease of processing. However, a significant drawback is its hydrophobic nature, which poses issue with cell adhesion and viability. PDMS also adsorb small hydrophobic molecules, potentially interfering with drug testing by skewing results. Additionally, PDMS requires an expensive clean-room facility for master mold fabrication, and is not easily adaptable to large-scale commercial production.
To address these challenges, we developed a 3D-printed lung Microphysiological system (MPS) with an integrated ECM that could effectively mimic the in-vivo 3D-microenvironment of lung tissue. The designed platform features true 3D-demintionality, facilitating ease prototyping, and scaling up, while avoiding the adsorption of small hydrophobic molecules. Additionally, the actuation induces radial stretching of ECM similar to alveolus-known as radial traction. More interestingly, the chip has advantages over other (PDMS) due to its ECM membrane made of collagen-elastin, making it more physiologically relevant. 3D-printed lung MPS comprises of two chambers separated by the ECM membrane, and an actuating membrane (Fig.1). The device design was conceptualized using CAD software and printed with SLA 3D-printer using Next-DentOtho Clear resin. The printed devices were washed with isopropyl alcohol and cured under UV for one hour. The ECM membrane was synthesized with 1:1molar of Collagen and Elastin (4mg/mL). DYMAX® MD UV-curable adhesive was used to attach ECM membrane and tubing to the device.
Joshua Whiting (co-authors: Audri Dara, James Kwan, Anna Edmunds, Sheri Holmen, and Jan Kubanek)
Faculty Advisor: Jan Kubanek
Title: Targeted drug release for effective treatment of glioblastoma (GBM)
Abstract: Glioblastoma (GBM), the most lethal primary brain tumor, faces treatment challenges due to the blood-brain barrier’s (BBB) tight junctions restricting drug delivery. Systemic administration of potent chemotherapeutics like doxorubicin causes severe off-target toxicities, limiting both efficacy and safety. Focused ultrasound (FUS)-mediated BBB disruption offers promise but lacks spatiotemporal control over drug release. To address this, we developed polymer-encapsulated doxorubicin microbubbles (~2 µm) for FUS-triggered delivery. These microbubbles combine PDLLA-mPEG (a diblock copolymer of lactic acid and ethylene glycol) for containment and biocompatibility and Perfluoro-octyl-bromide (PFOB) for acoustic responsiveness, enabling localized drug release in conjunction with temporary BBB disruption, while minimizing systemic exposure. This study evaluates survival benefits in a clinically relevant GBM mouse model.
Ava Yektaeian Vaziri (co-authors: Kyoichiro Yazaki, Emmanuel Offei, Sofia Ruiz Castillo, Muhammad S Khan, Ankur Shah, Christopher Runge, Douglas Smego and Derek J. Dosdall)
Faculty Advisor: Derek J. Dosdall
Title: Telemetry Reveals Arrhythmia Onset and Resolution in the Days Following Heart Attack in a Preclinical Model
Abstract: Heart failure (HF) remains a major cause of morbidity and mortality worldwide, and preclinical models are essential for studying HF progression. Left anterior descending (LAD) coronary artery ligation is a well-established method for inducing chronic ischemic heart failure, yet early postoperative arrhythmic patterns remain poorly characterized. During the development of a preclinical heart failure model, we observed persistent episodes of accelerated idioventricular rhythm (AIVR) shortly after LAD ligation. AIVR is a ventricular arrhythmia defined by three or more consecutive wide-complex ventricular beats at a heart rate typically between 50 and 160 beats per minute, often resulting from increased automaticity of Purkinje fibers and associated with atrioventricular (AV) dissociation. Although AIVR is considered benign and self-resolving, little is known about its duration, resolution timeline, or morphological variability after myocardial infarction in preclinical animal models.
BMES 2026 will be held in October, 2026 in Orlando, FL.